Abstract
Background: Despite maximally-intensive chemotherapy and stem cell transplantation, survival of patients with the very rare t(17;19)/TCF3-HLF B-acute lymphoblastic leukemia (B-ALL) subtype remains effectively 0%. Prior studies have demonstrated association of the oncogenic TCF3-HLF fusion protein with multi-drug resistance via increased expression of ABC and P-glycoprotein drug efflux transporters, as well as via upregulation of pro-survival Ras and BCL-2 pathways. Preclinical studies and small clinical case series of targeted inhibitor addition to chemotherapy or antibody-based and cellular immunotherapies have aimed to improve outcomes for children with TCF3-HLF ALL. Unfortunately, targeting of these activated pathways with the BCL-2 inhibitor venetoclax or other small molecule inhibitors (SMIs) has been minimally or only transiently effective, suggesting more complex mechanism(s) of chemoresistance. In recent years, many patients with relapsed TCF3-HLF ALL have enrolled on clinical trials of CD19- or CD22-targeted immunotherapies. However, TCF3-HLF ALL frequently harbours deactivating mutations in PAX5, a major B-cell regulator and indispensable CD19 transcription factor, placing immunotherapy-treated patients at higher risk of CD19 antigen-loss relapse. New therapies remain needed to prevent relapse and attempt cure.
Methods: We designed an unbiased kinome-wide CRISPR/Cas9 library to identify essential drivers in TCF3-HLF leukemogenesis. We screened the human TCF3-HLF ALL cell line HAL-01 and our TCF3-HLF ALL patient-derived xenograft (PDX) model ALL1807 (Hurtz JCI 2020, Schultz Genome Biol 2021), then validated identified targets using 49 SMIs targeting receptor tyrosine kinases (RTK), MEK signaling, and cell cycle pathways. We selected promising candidate inhibitor pairings with non-overlapping mechanisms of action and assessed for in vitro drug synergy via SynergyFinder analyses. Finally, we assessed the in vivo activity of targeted inhibitors in ALL1807 and two newly established TCF3-HLF ALL PDX models (CPCT-0002, CPCT-0003) created from primary pediatric specimens obtained via the LEAP Consortium (Pikman Cancer Disc 2021).
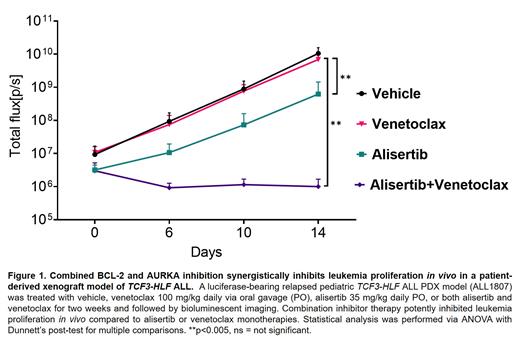
Results: RNA-sequencing of HAL-01 and ALL1807 cells followed by functional protein association (STRING) analysis confirmed a network of significantly upregulated (>3-fold) plasma membrane and cytoplasm components of RTK pathways as well as BCL-2. The intersection of the results of the SMI drug library screen with the top 1% targets identified in CRIPSR/Cas9 screen determined p120-RasGAP and Aurora kinase A (AURKA) as therapeutic targets in TCF3-HLF ALL. In vitro treatment of HAL-01 or ALL1807 cells with the RasGAP inhibitor, pluripotin, or the AURKA inhibitor, alisertib, across a range of concentrations demonstrated robust anti-ALL activity. AURKA and RasGAP co-immunoprecipitated and this protein complex was disrupted with alisertib or pluripotin treatment. The AURKB inhibitor barisertib had minimal activity against TCF3-HLF ALL cells, confirming preferential dependency of these cells upon AURKA. Treatment of TCF3-HLF ALL cells with the BCL-2i venetoclax did not disrupt the AURKA/RasGAP complex, suggesting its different mechanism of action and potential for combinatorial drug therapy. Next, we found that alisertib and venetoclax synergistically killed TCF3-HLF ALL cells. Finally, we observed superior inhibition of in vivo leukemia with dual AURKA and BCL-2 inhibitor treatment of three TCF3-HLF ALL PDX models compared to single-agent alisertib or venetoclax (Figure 1).
Conclusions: We identified AURKA as a critical new driver in TCF3-HLF ALL via orthogonal genetic and functional assays and confirmed prior observations of BCL-2 dependency in our models. We validated these key targets via in vitro and in vivo pharmacologic inhibition studies with drug synergy detected with combined alisertib and venetoclax in human TCF3-HLF ALL cell lines and PDX models. We posit that dual AURKA and BCL-2 inhibition is a clinically-pragmatic and potentially effective therapeutic strategy for patients with this rare, but highly fatal, leukemia subtype that merits formal clinical investigation.
Carroll: Incyte Pharmaceuticals: Research Funding; Janssen Pharmaceutical: Consultancy. Stegmaier: Auron Therapeutics, Kronos Bio, AstraZeneca, Novartis Institute of Biomedical Research: Consultancy, Research Funding. Tasian: Incyte Corporation: Research Funding; Gilead Sciences: Research Funding; Kura Oncology: Consultancy; Aleta Biotherapeutics: Consultancy.